Abstract
Purpose:
Helicobacter pylori is one of the most prevalent infectious agents in the world causing a variety of gastrointestinal diseases including gastritis, peptic ulcer and gastric carcinoma. The objective of this study was to comparatively evaluate invasive tests (rapid urease test and polymerase chain reaction) and non-invasive tests (enzyme-linked immunosorbent assay) PCR-und-ELISA-kits in the diagnosis of mycotoxigenic H. pylori infection.
Methods:
Biopsy samples and sera were collected from 105 patients with gastric disorders. The presence of H. pylori infection in gastric biopsies was evaluated by RUT and PCR methods using chemotaxis signal transduction protein (CSTP) gene primers, Urea C and HP-16srRNA. Serum samples were used for the ELISA test. Detection of infection with cag A positive strains was performed by PCR and the cag A-IgG ELISA kit.
Results:
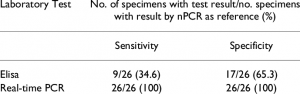
Patients with at least two of three positive results were considered infected. The sensitivity, specificity, predictive value and precision of the three different methods were evaluated. Of the 105 gastric biopsies, H. pylori were positive in 51 patients (48.57%). The best sensitivity (92.16%) corresponded to the RUT. The sensitivities of other tests, including the PCR and ELISA tests, were 88.24% and 90.20%, respectively. The PCR showed the best specificity (94.44%), and the specificities of the other tests, including the RUT and ELISA tests, were 90.74% and 61.11%, respectively. In addition, the results of the PCR and cag A-IgG ELISA showed a high prevalence of the positive cag A strain in the study population.
Conclusion:
According to our findings, ELISA in serum is a rapid non-invasive test for the detection of H. pylori infection in the absence of an indication for endoscopy. Furthermore, considering the high prevalence of mycotoxigenic strains of H. pylori, cag A is suggested as a promising target for non-invasive PCR and ELISA tests for the detection of infections with toxigenic strains.
Keywords:
Helicobacter pylori, polymerase chain reaction, enzyme-linked immunosorbent assay, Cag A, rapid urease test